Serum Institute investing USD 100 million on potential COVID-19 vaccine
Our manufacturing facility is ready and we plan to start production in two months. We are spending more than USD 100 million for this facility. Till the trials are completed successfully for safety and efficacy, vaccines will not be distributed either in India or anywhere else in the world," Adar Poonawalla, CEO, SII, said.
PUNE: Serum Institute of India, the world's largest vaccine manufacturer, is investing USD 100 million on a potential COVID-19 vaccine being developed at Oxford University, according to a company official. The Pune-based company has partnered with AstraZeneca, a British biopharma giant, to ensure equitable supply of the AZD1222 vaccine doses to India as well as low-and-middle-income countries.
"Our manufacturing facility is ready and we plan to start production in two months. We are spending more than USD 100 million for this facility. Till the trials are completed successfully for safety and efficacy, vaccines will not be distributed either in India or anywhere else in the world," Adar Poonawalla, CEO, Serum Institute of India (SII), said.
"Having said that we will start making a few millions of doses and stockpiling it at personal risk," added Poonawalla, while plying to a question on the firm's plans to move ahead in terms of production of the doses.
To a question on how much doses India is likely to get in the first phase and by when they would be available, he said that it is a bit early to comment on a specific number.
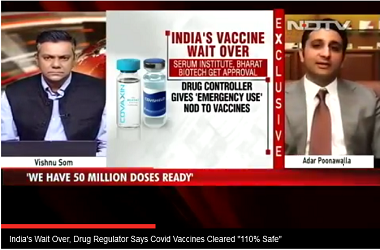
"However, if the vaccine trials succeed, India will gain access to the doses as it will also be the requirement of the Government of India. And we are certain that everybody will respect if the substantial volumes go to India," he added.
Earlier, Poonawalla, on tie-up with the British drug major had said, "SII is delighted to partner with AstraZeneca in bringing this vaccine to India as well as low-and-middle-income countries."
Over the past 50 years, SII has built significant capability in vaccine manufacturing and supply globally. "We will work closely with AstraZeneca to ensure fair and equitable distribution of the vaccine in these countries."
Talking about SII's endeavour to develop a vaccine with USA based firm Codagenix, Poonawalla recently said that at present, the firm is undergoing pre-clinical trials (animal trial phase) and hope to progress to the human trial phase by September/October.
"The aim is to make the vaccine over the next 1.5-2 years to help combat the novel coronavirus. With the combined efforts of our partner Codagenix, we are optimistic that we will be able to provide a viable and effective vaccine for mass use," he had said.
Besides, Oxford and Codagenix, the firm is also working with Austria's Themis and other two vaccine candidates.
At least half a dozen attempts at creating a vaccine against coronavirus are on globally and the efforts at Oxford are considered one of the most promising.
A vaccine will be a radical intervention which can arrest the loss of lives due to the infections.
Source - ET HealthWorld
ABOUT US
OUR COMPANIES
CORPORATE SOCIAL RESPONSIBILITY
© Copyright 2026. Cyrus Poonawalla Group. All rights reserved.